Hey everyone, I need to talk about something really serious that’s happening right now in Telangana. On January 10, 2026, the Drugs Control Administration issued an urgent stop-use notice for Almont-Kid Syrup, and the reason is genuinely alarming. This children’s medicine, which should have been safe, was found contaminated with ethylene glycol – a highly toxic substance.
This is not a small quality issue. Ethylene glycol poisoning is a medical emergency, and even small amounts can cause life-threatening kidney damage, especially in children.
Let’s break down what happened, what ethylene glycol is, how it harms the body scientifically.
What Exactly Happened?
Telangana DCA clarified that they received an alert from CDSCO (Central Drugs Standard Control Organization), East Zone, Kolkata, based on a laboratory report that declared the syrup adulterated with Ethylene Glycol.
The stop-use notice specifically mentions:
- Product: Almont-Kid Syrup (Levocetirizine Dihydrochloride + Montelukast Sodium)
- Batch No.: AL-24002
- Mfg.: Jan-2025
- Exp.: Dec-2026
- Manufacturer: Tridus Remedies, Hajipur, Bihar
Telangana’s instructions were strict:
- Public should stop using it immediately
- Retailers/distributors/hospitals should freeze stock
- People should report possession to DCA Telangana
- This information is from the official Telangana DCA press note.
Telangana DCA Official Press Note PDF
What is Ethylene Glycol?
Let me break down the chemistry for you –
Ethylene glycol (EG) is a small organic compound belonging to the diol (glycol) family. Chemically, it is known as:
- IUPAC name: Ethane-1,2-diol
- Molecular formula: C₂H₆O₂
- Molar mass: 62.07 g/mol
- Structure: HO–CH₂–CH₂–OH (a 1,2-diol)

Chemical Nature
It contains two hydroxyl (-OH) groups, making it a bifunctional alcohol, which is the main reason behind its strong hydrogen bonding behavior and high boiling point.
Physical properties
- Color: colorless
- Odour: almost odourless
- Taste: sweet (this makes it extra dangerous—kids may swallow it)
- Boiling point: ~197°C
- Density: ~1.11 g/mL
Why it is used in industry
Ethylene glycol is not a medicine ingredient — it’s an industrial chemical, mainly used as:
- Antifreeze / coolant
Because EG reduces the freezing point of water (colligative property), it is used in car radiators. - Polymer industry
It is a key monomer for making polyethylene terephthalate (PET):
EG + Terephthalic acid → PET (polyester plastics, bottles, fibers)
Why EG is “highly toxic” (Actual Chemistry)
Here’s the crucial chemistry part:
Ethylene glycol itself is not the main killer.
It becomes toxic after biotransformation in liver.
Ethylene glycol (EG) undergoes oxidation by Alcohol Dehydrogenase (ADH):
- Ethylene glycol (HO-CH₂-CH₂-OH)
→ (ADH) → Glycolaldehyde (HOCH₂CHO) - Glycolaldehyde
→ (ALDH) → Glycolic acid (HOCH₂COOH) - Glycolic acid
→ Glyoxylic acid (CHO-COOH)
→ Oxalic acid (HOOC-COOH)
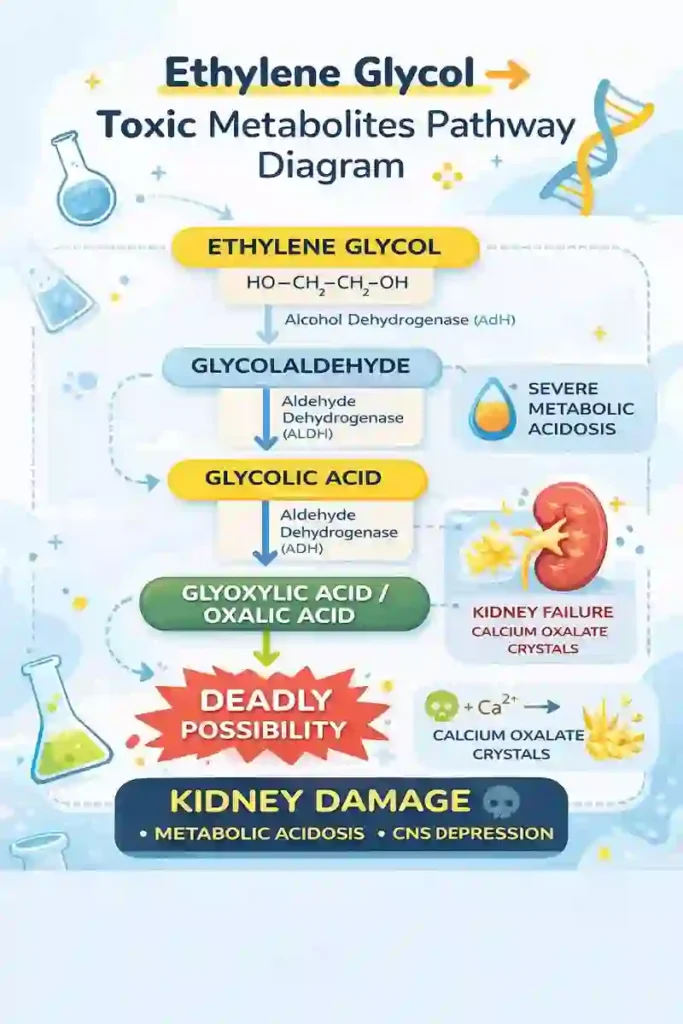
The Calcium Oxalate Crisis:
Here’s the killer mechanism: oxalic acid immediately complexes with calcium ions (Ca²⁺) in your bloodstream to form calcium oxalate monohydrate crystals (CaC₂O₄·H₂O). These needle-shaped crystals precipitate out of solution and deposit in renal tubules, causing direct tubular necrosis. Under polarized light microscopy, you’d see the characteristic envelope-shaped or prismatic birefringent crystals in urine sediment – a diagnostic hallmark of ethylene glycol poisoning.
Oxalic acid + Ca²⁺ → CaC₂O₄ (calcium oxalate crystals)
Main toxic reasons:
✅ Glycolic acid causes severe metabolic acidosis
✅ Oxalic acid binds Ca²⁺ → forms calcium oxalate crystals
These crystals deposit in kidney tubules → leading to:
- acute tubular necrosis
- renal shutdown
Why it should NEVER be in syrups
In pharmaceutical chemistry:
- oral liquid medicines use excipients like glycerin, propylene glycol, or sorbitol
- EG is cheaper but toxic
- EG contamination indicates raw material fraud or bad quality control
So if ethylene glycol is detected in a syrup, regulators treat it as:
✅ adulteration and serious public health emergency
Why Ethylene Glycol Is So Dangerous for Children
A child’s body is small… soft… still developing.
So when a toxic chemical like ethylene glycol enters their body, the damage doesn’t take days, it can start within hours.
- It turns the child’s blood acidic (Metabolic acidosis)
- It silently destroys kidneys (Most dangerous part)
- It affects the brain & nervous system
Why children are at higher risk than adults
This is a painful truth:
- children have low body weight
- even a small contaminated dose becomes “high dose”
- kidney damage develops very quickly
- dehydration makes poisoning worse
Is this article helpful?
Stay Safe and Stay Informed
This is exactly why we need to stay informed about public health alerts and take them seriously. The government has taken swift action, but our vigilance as consumers and parents is equally important. Always purchase medicines from licensed pharmacies, check batch numbers when alerts are issued, and never hesitate to report suspicious products.
If you found this information helpful, please share it with friends and family in Telangana and across India. Awareness can literally save lives.
Stay safe, everyone.
For official information and updates, contact the Drugs Control Administration, Telangana, or visit their official website. In case of emergency or suspected poisoning, call your local poison control center or go to the nearest emergency room immediately.
Sources:
- Telangana DCA Official Press Note PDF
- NCBI Bookshelf (StatPearls): Ethylene Glycol Toxicity
- PubMed Central (2025): Ethylene glycol poisoning complicated by cardiac arrest and organ damage
Q. Is it true that Telangana banned Almont-Kid Syrup?
Not exactly.
The official notice is a “Stop Use Notice”, mainly for a specific batch (Batch No: AL-24002) found adulterated with Ethylene Glycol.
So it’s more of a batch-level safety alert, not necessarily a full permanent ban on all batches.
Q. What is the official batch number mentioned in the notice?
The press note clearly mentions:
Batch No.: AL-24002
Mfg Date: Jan 2025
Exp Date: Dec 2026
If your syrup has this batch number → do not use it.
Q. What is Ethylene Glycol (EG)?
Ethylene glycol is a toxic industrial chemical, commonly used in:
antifreeze/coolant
industrial solvents
⚠️ It is NOT safe for human consumption — especially for children.
Q. Why is Ethylene Glycol dangerous to health?
Because inside the body, Ethylene Glycol gets converted into toxic acids (like glycolic acid and oxalic acid).
These can cause:
severe acidity in blood (metabolic acidosis)
damage to kidneys (kidney failure)
breathing difficulty
seizures / coma in severe poisoning
Q. Is the syrup itself a “fake medicine”?
The notice says the syrup batch was adulterated, meaning:
The medicine may be real, but contaminated with a dangerous chemical. That’s why regulators issue stop-use notices.

I’m Mehebub Alam Chowdhury, an M.Sc. Organic Chemistry student, and my mission with Decodepure is to simplify complex chemical ingredients in everyday products. With my knowledge of chemicals, I aim to help you make safer, healthier choices by breaking down product labels in an easy-to-understand way.










